Lastly, we will consider general mechanisms for signal transduction across membranes of any cell that
must respond to its environment. Typically the agent that signals a cell to respond is a molecule (or in the case of light
sensation a photon) which binds either to a cell surface receptor or to a cytoplasmic receptor if the signaling agent is hydrophobic.
In almost all cases, such signaling activates protein kinases in the cell. Kinases are a class of enzymes which
use ATP to phosphorylate molecules within the cell.
The names given to kinases shows the substrate which is phosphorylated by the enzyme. For example:
- Hexokinase - an enzyme that uses ATP to phosphorylate hexoses.
- Protein kinase - enzymes that use ATP to phosphorylate proteins
within the cell. (Note: Hexokinase is a protein, but is not a protein kinase).
- Phosphorylase Kinase: an enzyme that use ATP to phosphorylate
the protein phosphorylase within the cell
In this guide we will review the activation by extracellular signals of 5 different protein kinases within
the cell. These kinases phosphorylate other proteins within the cell and through associated conformational and charge changes,
the phosphorylated proteins are either activated or inhibited in the expression of biological activity.
If a protein is phosphorylated by a kinase, the phosphate group must eventually be removed by a phosphatase
through hydrolysis. If it wasn't, the phosphorylated protein would be in a constant state of either being activated or inhibited.
Kinases and phosphatases regulate all aspects of cellular function. Some people estimate that 1-2% of the entire genome may
encode kinases and phosphatases. If so, there might be as many as 800 different kinases and phosphatases. We will review
only the properties of five major protein kinases which are activated through interactions with different ligands. The mechanisms of activation
of the different types of kinases will be described below.
Figure: five major protein kinases
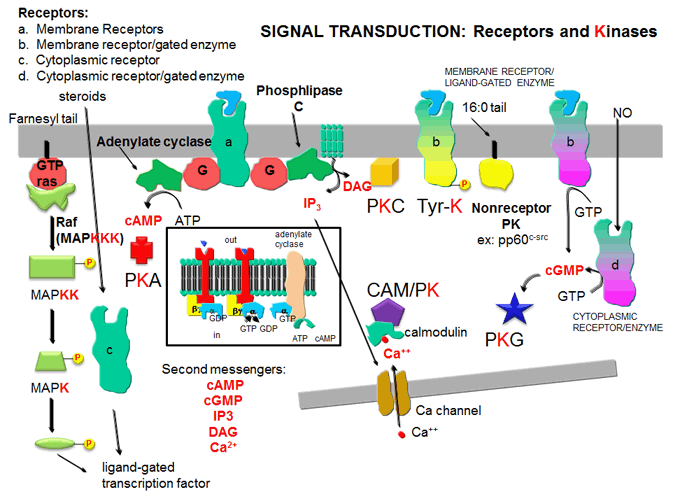
KINASES AND THEIR ACTIVATION
1. Protein Kinase A (PKA): Cascade of events: A transmembrane receptor
WITHOUT ENZYMATIC ACTIVITY binds an extracellular chemical signal, causing a conformational change in the receptor which propagates
through the membrane. The intracellular domain of the receptor then binds to an intracellular heterotrimeric G protein
(since it binds GDP/GTP) in the cell. The G protein dissociates and one subunit interacts with and activates an enzyme
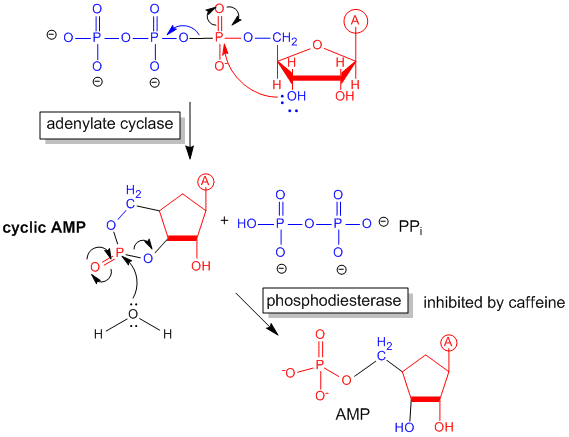
- adenylate cyclase- which converts ATP into a second messenger - cyclic AMP (cAMP)
- in the cell. cAMP activates protein kinase A (PKA)
which phosphorylates proteins at specific Ser or Thr side chains.
Figure: cyclic AMP
Receptors which work through an intermediary G protein usually are single polypeptide chains that span
the membrane seven times in a serpentine fashion.
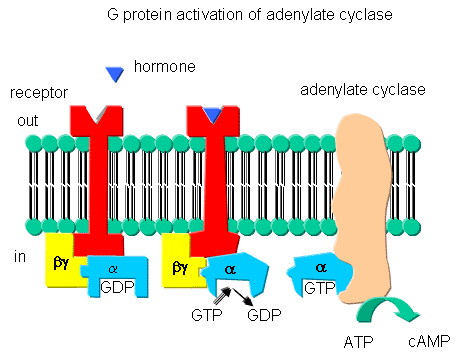
Figure: G PROTEINS AND ADENYLATE CYLASE ACTIVATION
Some signals that activate adenylate cyclase and use cAMP as a second messenger include: corticotrophn,
dopamine, epinephrine (b-adrenergic), follicle-stimulating hormone, glucagon,
many odorants, prostaglandins E1and E2, and many tastants.
Some enzymes regulated by cAMP-dependent phosphorylation by PKA
| Enzyme |
Pathway |
| Glycogen Synthase |
glycogen synthesis |
| Phosphorylase Kinase |
glycogen breakdown |
| Pyruvate Kinase |
Glycolysis |
| Pyruvate Dehydrogensae |
Pyruvate to acetyl-CoA |
| Hormone-sensitive Lipase |
Triacylglyeride breakdown |
| Tyrosine Hydroxylase |
Synthesis of DOPA, dopamine, norepinephrine |
| Histone H1 |
Nucleosome formation with DNA |
| Histone H2B |
Nucleosome formation with DNA |
| Protein phosphatase 1 Inhibitor 1 |
Regulation of protein dephosphorylation |
| CREB |
cAMP regulation of gene expression |
| PKA cosensus sequence |
XR(R/K)X(S/T)B (B = hydrophobic amino acid) |
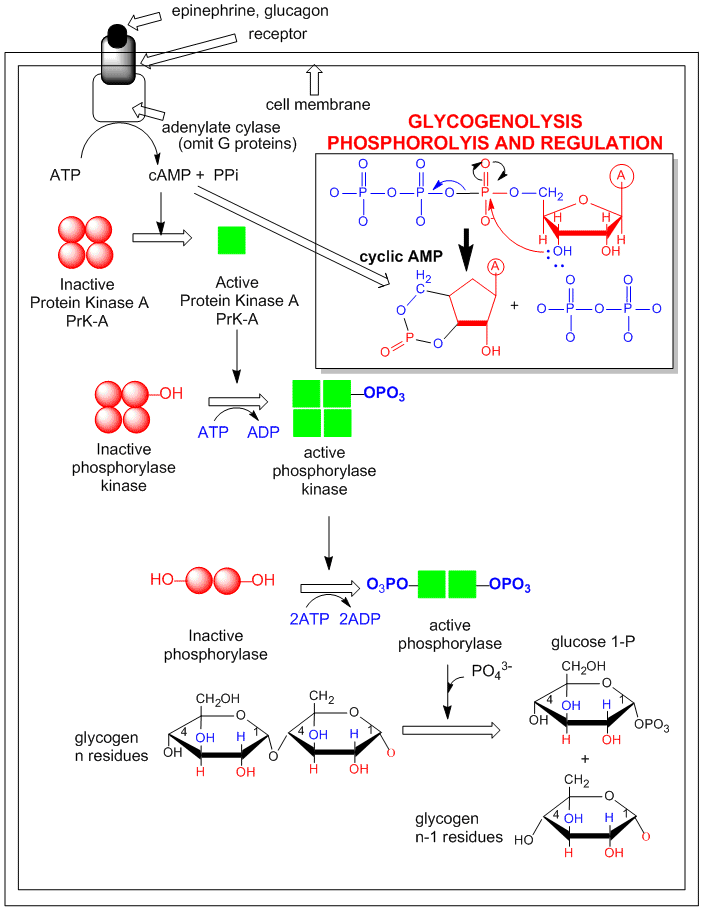
An example of how epinephrine (a flight/fight hormone) can lead to breakdown of glycogen (your main carbohydrate
reserves in muscle and liver) is shown below. A cascade of events, starting with the binding of the hormone to its receptor,
followed by activation of adenylate cyclase, which forms cAMP, which activates PKA, which leads to the activation of the enzyme
that breaks down glycogen (glycogen phosphorylase) is shown. (For simplicity, G protein involvement is not shown.)
Figure: Activation of glycogen phosphorylase through activation of PKA.
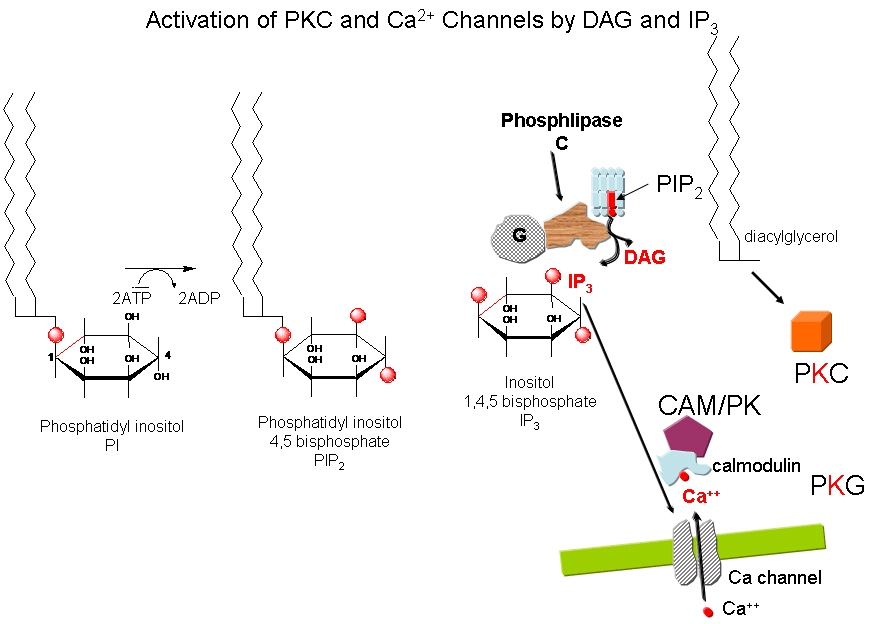
2,3. Protein Kinase C (PKC) and calmodulin-dependent
kinase (CAM-PK): Cascade of Events: A transmembrane receptor WITHOUT ENZYME ACTIVITY binds
an extracellular chemical signal, causing a conformational change in the receptor which propagates through the membrane.
The intracellular domain of the receptor then binds to an intracellular heterotrimer G protein (since it binds GDP/GTP) in
the cell. The G protein dissociates and one subunit interacts with and activates an enzyme - phospholipase
C - which cleaves the phospho-head group from a membrane phospholipid - phosphatidyl inositol - 4,5-bisphosphate
(PIP2) into two second messengers - diacylglyerol and inositol trisphosphate (IP3). Diacylglycerol
binds to and activates protein kinase C (PKC). The IP3 binds to ligand-gated
receptor/Ca++ channels on internal membranes, leading to an influx of calcium ions into the cytoplasm. Calcium
ions bind to a calcium modulatory protein, calmodulin, which binds to and activates the calmodulin-dependent kinase (CAM-PK). The released calcium ions also activate
PKC. As in the previous example, these receptors which interact with G proteins are single polypeptide chains which contain
7 membrane spanning alpha helices. The cycle of degradation and resynthesis of PIP2 is called the PI cycle.
Figure: PI cycle
Some signals that activate phospholipase C and make IP3 and diacylglycerol include: acetylcholine
(a different class than the type located at the neuromuscular junction that we discussed in the last chapter section), angiotensin
II, glutamate, histamine, oxytocin, platelet-derived growth factor, vasopressin, gonadotropin-releasing hormone, and thyrotropin-releasing
hormone. Some proteins phosphorylated by PKC include
Some kinases regulated by calcium and calmodulin include: myosin light chain kinase, PI-3 kinase,
CAM-dependent kinases. Ca/CAM also regulates other proteins which include: adenylate cyclase (brain), Ca-dependent Na
channel, cAMP phosphodiesterase, calcineurin (phosphoprotein phosphatase 2B), cAMP gated olfactory channels, NO synthase,
and plasma membrane Ca/ATPase.
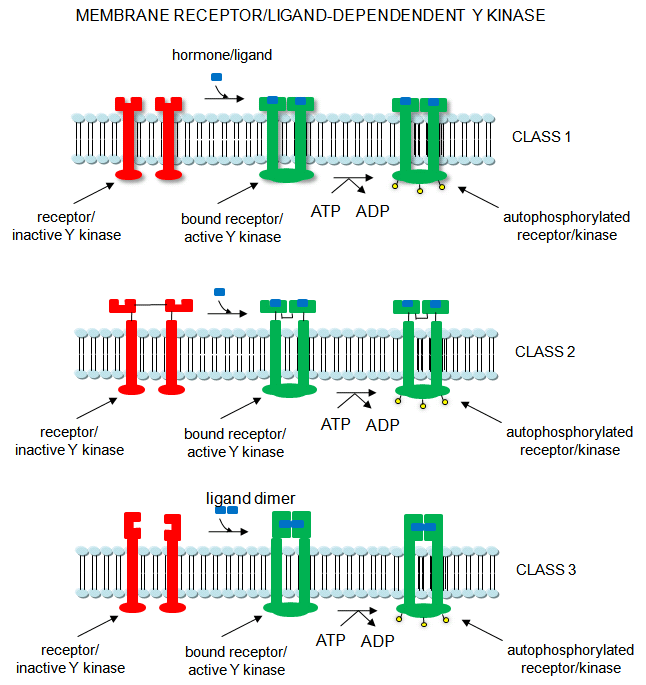
4. Receptor Tyrosine Kinases: Cascade of events: A transmembrane receptor WITH
HORMONE-DEPENDENT ENZYMATIC ACTIVITY (tyrosine kinase) binds an extracellular chemical signal, causing a conformational change
in the receptor which propagates through the membrane. The intracellular domain of the receptor becomes an active tyrosine
kinase which can phosphorylate itself (autophosphorylation) or other proteins. Such kinases are usually
active in a multimeric state. Typically, binding of two molecules of a ligand or a ligand dimer to individual subunits of
the receptor causes the monomers of the receptor to dimerize. In this form the kinase activity of the receptor is activated.
The individual subunits of the multimer are proteins with a single transmembrane helix. Examples are the insulin receptor
and epidermal growth factor receptor.
Receptor Tyr kinases autophosphorylate themselves, in a process required for their activity. When
the receptor is phosphorylated, other proteins can bind to the cytoplasmic domain of the receptor Tyr kinase where they are
phosphorylated. The target substrates phosphorylated by the receptor Tyr kinase are proteins with a common 100 amino
acid domain called SH for src homolgy, based on structural homology to another cytoplasmic protein, Src.
Src is an intracellular Tyr kinase activated when it binds through 2 SH domains to the autophosphorylated receptor Tyr kinase.
Specifically, the SH2 domain has been shown to bind phosphorylated peptides. These domains target proteins to the autophosphorylated
receptor Tyr kinase.
Figure: Receptor/Ligand-Dependent Protein Kinases
5. Protein Kinase G (PKG): Cascade of events: A transmembrane receptor OR a cytoplasmic
protein WITH LIGAND-DEPENDENT ENZYMATIC ACTIVITY (guanylate cyclase) binds an extracellular chemical signal (for the
transmembrane receptor) or an intracellular ligand (for the cytoplasmic protein receptor), causing a conformational change
in the receptor. The bound receptor is now a guanylate cyclase which converts GTP into cyclic GMP. This second messenger,
like cAMP which binds to and activates PKA, binds to and activates the cGMP-dependent protein
kinase (PKG). An example of a hormone which works through a transmembrane receptor is atrial naturetic
factor, which binds to a cell-surface receptor whose shape change is communicated to the intracellular receptor domain through
the single transmembrane helix, leading to the activation of the intracelluar guanylate cyclase domain of the protein.
Likewise, nitric oxide, NO, a gaseous ligand formed from Arg, can diffuse through the cell membrane, where it binds to the
heme of a cytoplasmic receptor/guanylate cyclase which form cGMP, leading to activation of PKG. (Steroid hormones also enter
the cell where they interact with a cytoplasmic receptor which often acts as steroid hormone-dependent transcription factor.
)
Kinases can be classified in many other ways. One is substrate specificity: Eukaryotes have
different kinases that phosphorylate Ser/Thr or Tyr. Prokaryotes also have His and Asp kinases but these are unrelated
structurally to the eukaryotic kinases. There are 11 structurally different families of eukaryotic kinases, which all
fold to a similar active site with an activation loop and catalytic loop between which substrates bind. Simple, single
cell eukaryotic cells (like yeast) have predominantly cytoplasmic Ser/Thr kinases, while more complex eukaryotic cells (like
human) have many Tyr kinases. These include the membrane-receptor Tyr kinases and the cytoplasmic Src kinases.
Recently, Manning et al. have analyzed the entire human genome (DNA and transcripts) and have identified 518 different
protein kinases, which cluster into 7 main families as shown in the chart below. Family membership was determined by
sequence comparisons of catalytic domains. They have named the entire repertoire of kinases in the genome the kinome.
Alterations in 218 of these appear to be associated with human diseases.
The Kinome
| Name |
Description |
| AGC |
Contain PKA, PKG, and PKC families |
| CAMK |
Ca2+/CAM-dependent PK |
| CKI |
Casein kinase 1 |
| CMGC |
Contain CDK, MAPK,GSK3, CLK famiies |
| STE |
homologs of yeast sterile 7, 11, 20 kinases |
| TK |
Tyrosine kinase |
| TKL |
Tyrosine kinase-like |
| others |
. |
Phosphatases
There are three main families of phosphatases, the phospho-Tyr phosphatases (PTP), the phospho-Ser/Thr phosphatases,
and those that cleave both. They can also be catagorized by the molecular sizes, inhibitors, divalent cation requirements,
etc. In contrast to kinases which differ in the structure of their catalytic domains, many phosphatases gain specificity
by binding protein cofactors which facilitate translocation and binding to specific phosphoproteins. The active phosphatase
hence often consists of a complex of the phosphatase catalytic subunit and a regulatory subunit. Regulatory subunits
for Tyr phosphatases may contain a SH2 domain allowing binding of the binary complex to autophosphorylated membrane receptor
Tyr kinases.
Important Ser/Thr phosphatases include:
-
Protein phosphatase 1 (PP-1) - different regulatory subunits target this to the liver glycogen particles
or muscle sarcoplasmic reticulum
-
Protein phosphatase 2A (PP-2A) - is a trimer with catalytic, regulatory, and a scaffolding structural
subunits.
-
Protein phosphatase 2B (PP-2B) - also called calcineurin or Ca2+/Calmodulin dependent protein phosphatase - It consists of a catalytic subunit (calcineurin A) and
a regulatory, calcium-binding subunit (calcinerin B). It is inhibited by the complex of the immunosuppressant cyclosporin
and FK506 with immunophilins. PP2B regulates PKA and PKC
-
Protein phosphatase 2C (PP-2C) -
PP1, 2A and 2B share a great deal of amino acid homology, and based on this homology, belong to one family.
PP2C belongs to another.
Protein Tyr phosphatases (PTPases) consist of receptor-like (transmembrane) and intracellular Tyr phosphatases.
They more resemble tyrosine kinases in their complexity than the Ser/Thr phosphatases. Important examples include:
-
PTP1B - dephosphorylates many cell surface receptors (insulin, EGF, PDGF) that have been phosphorylated
on Tyr residues. Its main activity seems to dephosphorylate nascent receptors in the endoplasmic reticulum before they
get to the final cell membrane destination.
-
Low molecular weight PTPase - These have roles in metabolism and differentiation of cells. They have a
molecular weight of 18,000 and have an active site CX5R-(S/T) motif, where the C (Cys) is an active site nucleophile.
Human adipocyte acid phosphatase is an example.
Web Links for Phosphatases
Receptors and G proteins
Receptors that interact with G proteins often have common characteristics. Consider the
b-adrenergic receptor, a prototype for such receptors. These bind epinephrine
and adrenaline, are found in muscle, liver, and fat cells, and are activated when energy reserves in these tissues are mobilized.
The receptors are single polypeptide which have 7 membrane-spanning a- helices.
Over 1000 similar serpentine receptor genes are found in humans, each encoding a protein of similar topology
but which bind different ligands. Many of the receptors bind to unknown ligands, and hence are called orphan
receptors. The unoccupied adrenergic receptor is associated with a heterotrimeric G protein, which contains an
a, b, and g subunits.
GDP is usually bound to the a subunit. When the hormone is bound to
the receptor, interactions of the receptor with the G protein (probably through the b and g subunits leads to conformational changes in the G protein
leading to replacement of GDP with GTP. This promotes dissociation of the a subunit
(with bound GTP), which is then free to bind to and activate adenylate cyclase, an adjacent membrane protein.
The a subunit is held to the membrane through a lipid anchor attached through
a post-translational modification. As long as GTP remains bound to the Ga subunit, it will continually activate adenylate cyclase. There needs to be a method to reverse this
process, otherwise no regulation of the process could occur. A built in regulatory mechanism does exist, since the
Ga subunit has GTPase activity. The GTP will eventually
hydrolyze, the GDP-Ga subunit will lose affinity for adenylate
cyclase, and return to for the heterotrimeric G protein associated with the unbound receptor.
Some bacterial toxins work by inactivating the GTPase activity of the Ga subunit, keeping it in the "stuck" position. For example, cholera toxin, an enzyme released by Vibrio
cholerae , catalyzes the ADP ribosylation of an Arg in the Ga
subunit by transferring everything but the nicotinamide from NAD+ to the Arg residue.
In contrast to the beta-adrenergic receptor, some Ga subunits actually inhibit adenylate cyclase when bound. These Ga subunits are called Gi in contrast to the stimulatory subunits, Gs. Some
Ga subunits interact with proteins other than adenylate cyclase.
We have already seen an example with the PKC activation of phospholipase C. There are many different Ga-like subunits expressed in different tissues.
Examples of different signals, receptors, Ga like-subunits, second messenger changes, and affected intracellular enzymes
| signal |
vasopressin |
epinephrine |
light |
odorant |
odorant |
sweet tastant |
| receptor |
VR |
b-adrenergic |
rhodopsin |
odorant recep. 1 |
odorant recept. 2 |
sweet receptor |
| Ga like-
subunit |
Gi |
Gs |
transducin |
Golfactory |
Golfactory |
Ggustatory |
| coupled enzyme |
adenylate cyclase |
adenylate cyclase |
phosphodiesterase |
phopholipase C |
adenylate cyclase |
adenylate cyclase |
| 2nd messenger |
decrease cAMP |
increase cAMP |
decrease cGMP |
increase IP3 |
increase cAMP |
increase cAMP |
| protein affected |
decrease PrK-A |
increase PrK-A |
dec. Ca, Na perm. |
inc. Ca perm |
inc.Ca, Na perm |
dec. K perm |
Another variant of a G protein is the protein ras. Mammalian cells contain
3 variants of ras: H, K, and N. They all bind GDP/GTP and have GTPase activity, and are members of
a large familty of small GTPase proteins. This protein is targeted to the cell membrane through the post-translational
addition of a hydrophobic farnesyl group. When activated by binding to GTP, it can bind a GAP protein (GTPase
activating protein) called Raf-1, which has tyrosine kinase activity.
The Receptome
There are an enormous variety of membrane receptor proteins in human cells. In analogy
to the genome, glycome, proteome, lipome and kinome, the human receptome has been shown to contain 20 different families containing
over 1000 different receptors.
Information about these receptors can be found at the Stanford Human Plasma Membrane Receptor web site.
Regulation by Phosphorylation
The activity of many different proteins (receptors, enzymes, channels) are regulated by phosphorylation
by kinases, and dephosphorylation by phosphatases. The most critical role for phosphorylation is probably
in the regulation of cell growth, division, and death. The cell cycle describes the various stages in the life of a
cell.
The cell cycle is divided into time intervals as shown in the diagram below. S stands for DNA synthesis,
M for mitosis (cell division), and G1 and G2 for gaps in between DNA synthesis which occurs in S and
mitosis which occurs in M. The diagram shows the cycle for a typical eukaryotic cell that is actively dividing.
Some cells enter a quiescent phase (Go) which represent a terminally differentiated cell, which may require months
to divide.
Figure: Cell Cycle
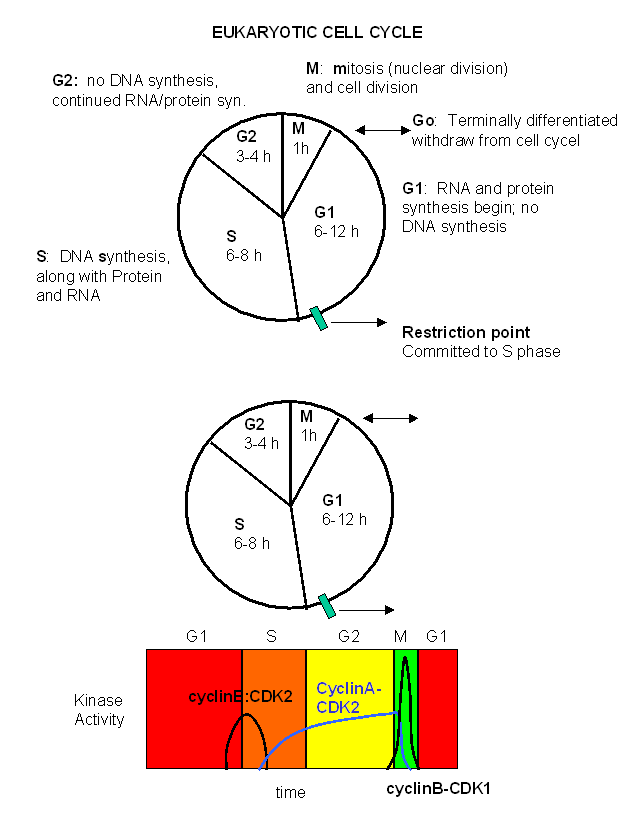
Passage through the cell cycle depends on the presence in the cell of two kinds of proteins:
- cyclins: which accumulate in the cell at various stage of the cell cycle and hence rise and fall
in cyclic fashion.
- cyclin-dependent protein kinases which form complexes with the cyclins to control their activity
For example, in frogs, cyclin dependent protein kinase 2 (CDK2) binds to cyclin B to form an active kinase
which phosphorylates a prereplication complex initiating S phase and mitosis. Cyclin B, a 45Kd protein, accumulates
to high levels just before S phase. Its concentration drops sharply at the end of mitosis. The kinase, a 34 Kd
protein, is encoded by the CDC2 gene (for cell division cycle gene).
A homologous gene exists in humans - the CDK2 gene (cyclin dependent kinase 2) - and controls entry in S phase.
These kinases can be considered heterodimers with a kinase catalytic subunit and a cyclin regulatory subunit. In animal
cells, there are at least ten different cyclins (A, B, .....) and at least eight different cyclin-dependent kinases (CDK1-8)
Another Look at Neurotransmission and Ion Channels
You may have noticed above that some signaling molecules, whose effects are regulated by kinases
(b-adrenergic and some olfactory signals by PKA and acetylcholine by PKC
for example), are neurotransmitters. In the previous chapter section, we discussed how neurotransmitters can act as
signals to open ion-specific, ligand-gated membrane channels, which change the transmembrane potential. In other words,
the neurotransmitters gate the channels directly. Typical examples of channels directly gated by neurotransmitters
are the acetylcholine receptor in neuromuscular junctions and the Glu, Gly, and GABA receptors in the central nervous system.
These receptors are multimeric proteins. Receptors with direct gating of ion flow are fast, with activities that last
milliseconds, and are used in eliciting behavioral responses.
However, ion channels can also be gated indirectly when the neurotransmitter binds to its receptor
and leads to events which open an ion channel that is distinct from the receptor. In this case, the occupied receptor
communicates to an ion channel indirectly through a G protein. Example of this indirect gating of ion channels include
the serotonin, adrenergic, and dopamine receptors in the brain. These receptors are classic single protein serpentine
receptors with 7 transmembrane helices, and intracellular domains that can interact with G proteins as described above to
increase second messenger levels (cAMP, DAG) in the neuron. These can either activate kinases in the cell, which phosphorylate
ion channels to either open or close them, or can bind directly to the channel and modulate its activity through an allosteric
conformational change. In some cases the G protein directly acts on the ion channel. These different ways
are described below. In contrast to direct gating, receptors that indirectly gate ion channels have activities that
are slow and last seconds to minutes. These receptors are usually involved in modulating behavior by changing the excitability
of neurons and the strength of neural connections, hence modulating learning and memory. These changes can occur in
many ways, summarized below and in the following link:
Figure: Neurotransmission: Gating through G Linked Receptors
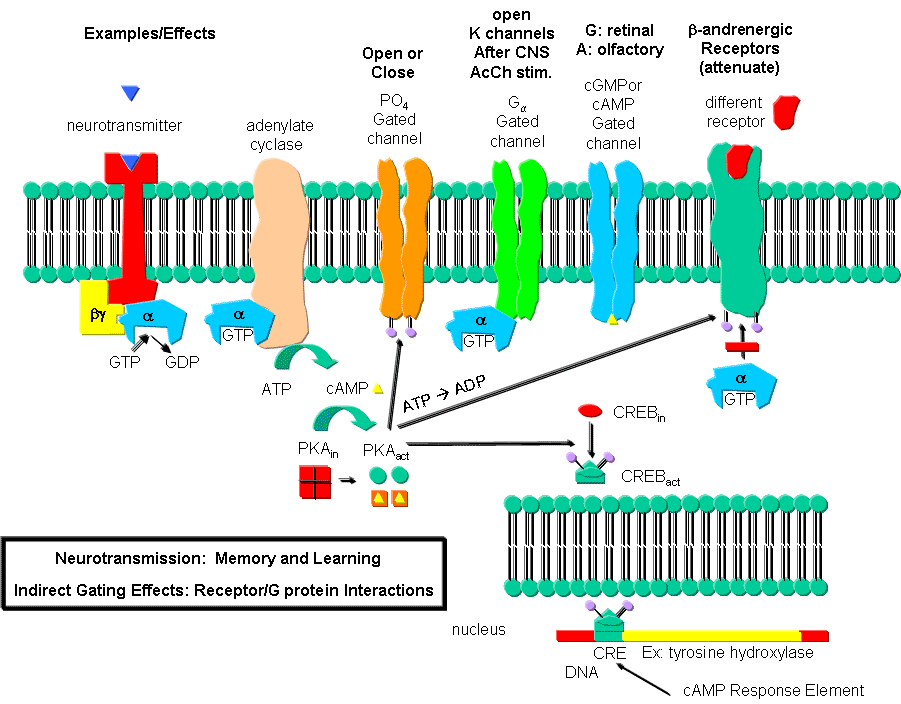
Phosphorylating ion channels: Receptors that act through a second messenger
system can change ion channel activity by activating kinases which phosphorylate the channels. This may:
-
open the channel normally closed at the resting potential and produce an effect like gating.
-
close a channel usually open at the resting potential (such as nongated K channels which when
closed would depolarize the cell and make it more excitable).
Ga interaction with ion
channels:
Second messenger interaction with ion channels:
Second messenger effects on proteins other than ion channels (usually different
receptors):
-
the b-adrenergic receptors are phosphorylated
by PKA and PKC (activated by stimulation of a different neurotransmitter receptor linked through a G protein to produce increased
levels of second messengers cAMP and diacylglycerol). When phosphorylated, the b-adrenergic receptor, itself activated through G protein) can't bind Gs. This in affect attenuates the response
of the b-adrenergic receptor to its own neurotransmitter which leads to desensitization
to that signal.
Second messenger regulates gene expression:
-
cAMP activated PKA can phosphorylate an inactive transcription factor in the cell, which then
can bind to a section of DNA called the cAMP Response Element (CRE), which is upstream of certain genes, leading to the transcription
of the genes. The transcription factor is called CREB for cAMP Response Element
Binding protein. Example: tyrosine hydroxylase (a monooxygenase) is involved in the synthesis
of epinephrine and norepinephrine. The activity of this protein is increased when it is phosphorylated by PKA.
Hence its activity can be increased quickly by this modification of the already present protein. If an animal is subjected
to severe or long term stress (cold or immobilization), presynaptic cells with norepinephrine will be stimlulated to release
the neurotransmitter. This requires continual synthesis of the neurotransmitter by the presynaptic cell.
The increase in synthesis of this neurotransmitter is caused by the presynaptic cell being stimulated by another neuron,
whch leads to increased levels of cAMP, and ultimately activation of CREB which increases transcription of the hydroxylase
gene.
Caffeine
Caffeine clearly produces a state of arousal in the central nervous system. High levels appear to block
the binding of an inhibitory neurotransmitter, adenosine, to the A2A adenosine receptor. In the absence of caffeine,
adenosine levels rise during the day, which promotes interaction with its receptor, leading to increasing sleepiness and lack
of concentration. When adenosine binds normally to its receptors, it activates the adenylate cyclase cascade,
which activates PKA, leading to changes in phosphorylation state of many proteins inside the cell, including protein phosphatase
(2A). These changes inhibit neural firing. Caffeine blocks these changes.
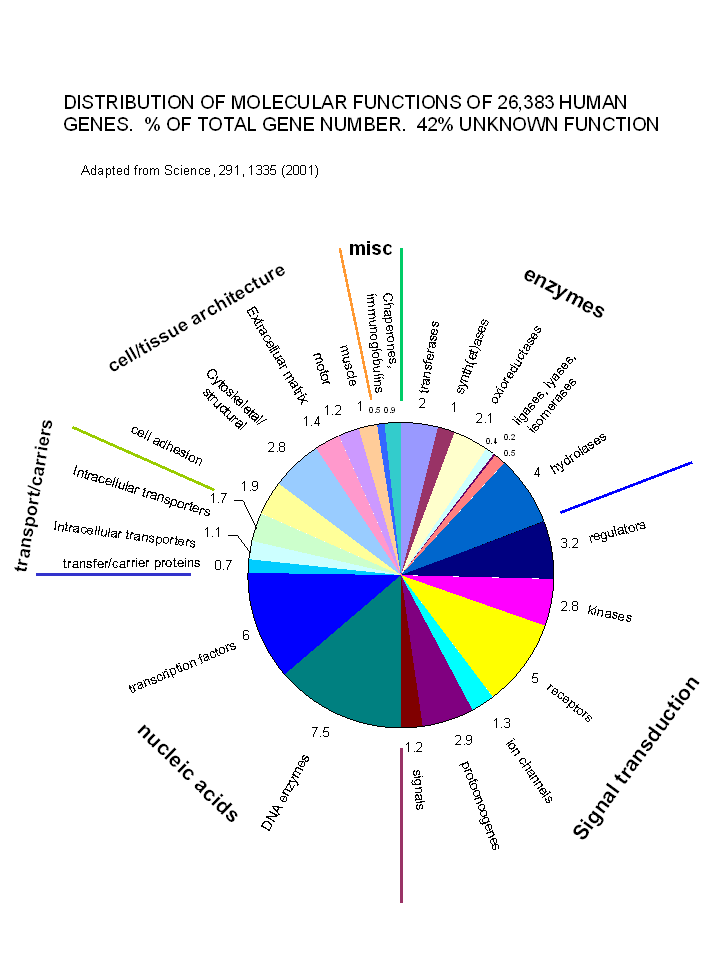
With the determination and annotation of the human genome, it has become very clear that a
significant fraction of the human genome (about 40% of the 58% of known genes determined by Venter et. al. and published in
Science, 291, 1335, 2001) is devoted directly or indirectly to signal transduction processes. These include signal molecules,
receptors, kinases, regulators, protooncogenes and ion channels. The chart below shows the relative distribution of
over 26,000 genes of known function (with 42% still of unknown function.
Figure: Distribution of Molecular Functions of 26,383 Genes (Does not show 42% of genes of unknown function)
Signal Transduction and Lipid Rafts
When extracellular signals bind to membrane receptors, conformational changes in the receptor
protein signals the inside of the cells that the receptor is bound with a ligand. Once bound, the receptor often move
in the membrane and clusters in outer leaflet rafts that contain cholesterol and spingholipids, with longer and more saturated
fatty acids. This increases the thickness and decreases the fluidity of the bilayer in the raft. Inner leaftlet
rafts are also observed. Caveolae (regions of the membrane that are invaginated) and to which the protein caveolin
is bound, are found in both leaftlets. Using fluorescent microscopy, Zacharias et. al. were able to detect movement
of specific proteins into raft regions. Proteins that are covalently modified with a fatty acid (myristic and palmitic
acid) move to the caveolae while isoprene-modified proteins (such as the the prenyl group geranylgeranyl) do not move to the
caveolae.
Web Sites
New Research
Schizophrenia is a complex brain disease that has been linked to alterations in neuronal brain signaling by many different neurotransmitters,
including serotonin (5HT), dopamine, and glutamate. Illicit drugs that also interact the receptors for those neurotransmitter
(LSD, amphetamne, and phenylcyclidine or PCP, respectively) also can induce symptoms characteristic of schizophrenia.
Might these three different drugs, and their cognate neurotransmitters, induce signal transduction mediated by a common protein?
Sveningsoon, Greengard, et al. have found such a common intermediary protein called DARPP-32, which regulates the activity
of signal transduction kinases/phosphatases in these different neurons.
To test their hypothesis that these drug signal produce effects which overlap at DARPP-32, they produced knock out mice
lacking this gene and investigated the effects of the drugs on these mice. Their "assay" was to look at alteration in
behaviors common to schizophrenic and some people on those drugs. In normal mice and people, if several "pulses" of
tactile stimulation are given before inducing a startle reflex, the reflex is inhibited. "Prepulse inhibition of a startle
reflex is inhibited in schizophrenics and in humans and mice under the influence of these drugs. Also, schizophrenics
and drug-treated mice/humans often repeat motions (perseverative behavior). Knock out mice did not experience these
behavior effects when given these drugs.
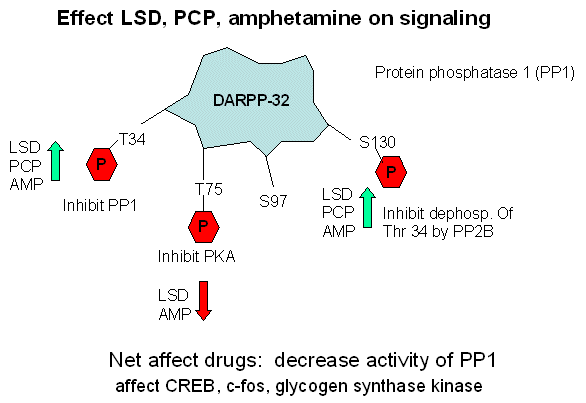
The DARPP-32 protein has four Ser/Thr residues which can be phosphorylated by kinases (at Thr 34, Thr 75, Ser 97, and Ser
130) and dephosphorylated by phosphatases. The phosphorylations each appear to have different effects:
- Thr 34 - inhibit protein phoshatase I (PP1)
- Thr 75 - inhibit protein kinase A (PKA)
- Ser 97 - activate protein kinase A (PKA) to phosphorylate Thr 34
- Ser 130 - prevent dephosphorylation of Thr 34 by protein phosphatase 2B (PP2B)
Sveningsson, Greengard, et al investigated the effects of LSD, amphetamine, and PCP on the phosphorylation state of these
amino acid side chains in DARPP-32. Their study showed that the ultimate effect of these drugs affect the phosphorylation
state of Thr 34, Thr 75, and Ser 130 in fashion that inhibits PP1 activity. (All three drugs increased phosphorylation
of Thr 34 and Ser 130, and amphetamine and LSD decreased Thr 75 phosphorylation.) The activity of proteins downstream
in the signaling cascade from PP1 (specifically glycogen synthase kinase 3 - GSK-30 , cAMP response element binding protein
- CREB) and c-fos) are then effected.
Figure: Effect LSD, PCP, and amphetamines on signaling