|
Properties of Membranes/Liposomes - Dynamics
Molecules are not static, but rather are dynamic. This also applies to molecular aggregates. Liposomes
and bilayers in general must be somewhat dynamics, otherwise they might be impenetrable barriers across which nothing could
pass. Cell membranes must separate the outside of a cell from the inside, but they must also allow passage of molecules and
even ions across the membrane. What is the evidence that membranes are dynamic?
First, lipids can diffuse laterally in the membrane. This can be shown as follows. Make a liposome from
phosphatidylethanolamine PE, which has been labeled with TNBS (Trinitrobenzensulfonate). The NH2 on the head group
of PE can attach the TNBS which undergoes nucleophilic aromatic substitution with the expulsion of the SO3-2.
The TNB group absorbs UV light. Next, using a fluorescent microscope, the fluorescence intensity of a region of the surface
can be recorded. Then shine a laser on a small area of the surface, which can photobleach the fluorescence in the area. Over
time, fluorescence can be detected from the region again. The rate at which it returns is a measure of the lateral diffusion
of the labeled lipids into the region. Lipids can undergo lateral diffusion at a rate of about 2 mm/s. This implies that the lipids can transit the surface of a bacteria in 1 sec.
Transverse, or flip-flop diffusion (movement of a phospholipid from one leaftlet to the other, not within a given leaflet) should be more difficult. Experimentally
this is investigated as shown in the diagrams below.
Liposomes experiments: To test flip-flop diffusion in an artificial membrane, liposome
are made with a mixture of PC and a PC derivative with a nitroxide spin label (has a single unpaired electron). Both inner
and outer leaflets of the membrane have the labeled PC. Like a proton in NMR spectroscopy, a single electron has a spin which
can give rise to an electron-spin resonance (ESR) signal (as a proton gives rise to a nuclear magnetic resonance signal) when
irradiated with the appropriate frequency electromagnetic radiation (microwave frequency for ESR, radio frequency for NMR)
in the presence of a magnetic field. The liposomes are kept at 0oC and the ESR signal is determined. Ascorbic acid,
a water soluble vitamin and antioxidant/ reducing agent, is added to the liposomes. This reduces the spin labeled PC in the
outer leaflet, but not the inner leaftlet of the bilayer since ascorbic acid can not enter the liposome or otherwise interact
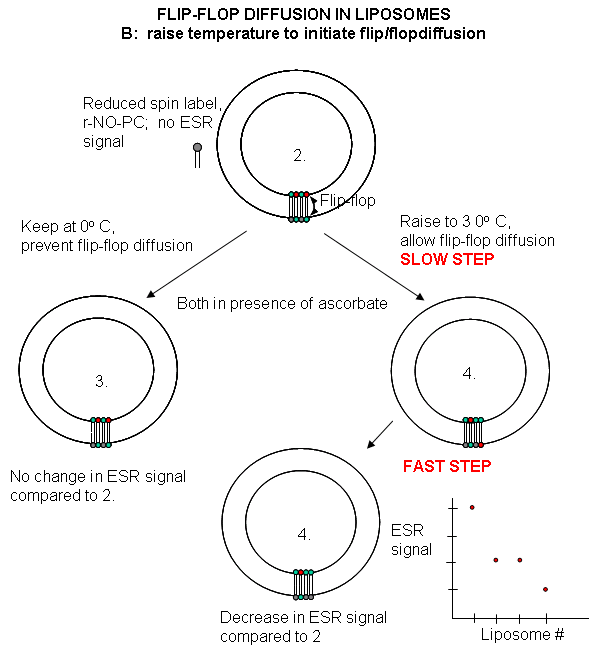
with it. This reduces the ESR signal to a lower, constant value. The sample is divided into two. One sample is left
at 0oC, the other is raised to 30oC. The ESR signal is recorded as a function of time. The 0oC
prep shows no change in ESR with time, while the 30oC prep ESR signal decreases with time. This decrease must result
from flip-flop diffusion of labeled PC from the inner leaflet to the outer, and subsequent reduction by ascorbic acid.
These experiments in experimental bilayer systems like liposomes shows that flip-flop diffusion is orders of magnitude slower
than lateral diffusion.
Figure: Flip/Flop diffusion in liposomes A: Making vesicles with ESR active PC analog
only in outer leaflet
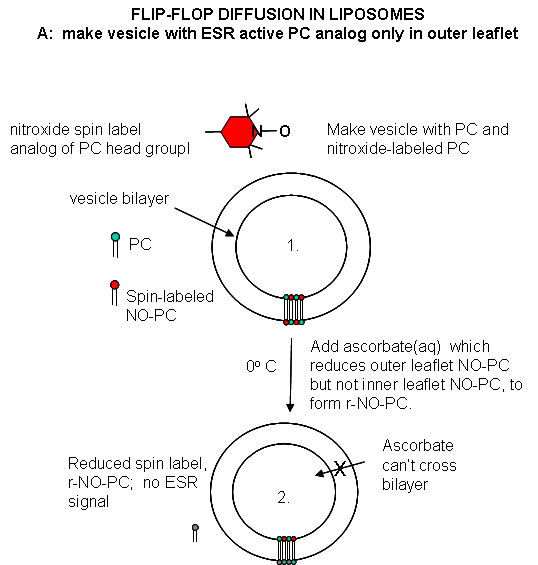
Figure: Flip/Flop diffusion in liposomes B: Raising
the temperature to initiate flip/flopdiffusion
Cell Experiments: An analogous experiment can be done with bacteria. Radiolabeled
32 PO4 is added to cells for one minute, which leads to the labeling of newly synthesized phospholipid
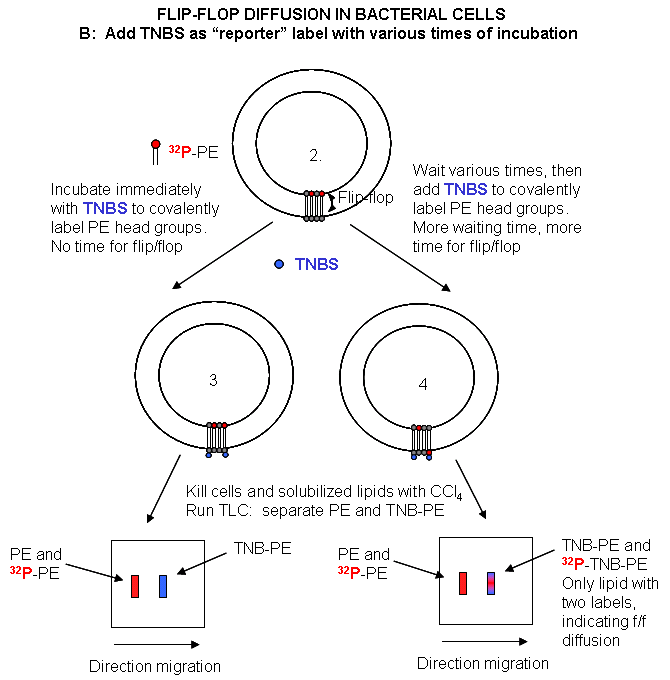
(PL) which locates to the inner leaflet. The cells are then split into two samples. One sample is reacted immediately
with TNBS, which will label only PE in the outer leaflet. The other sample is incubated 3 minutes (to allow PL synthesis)
and then reacted with TNBS. After a short labeling period, the cells are destroyed by adding organic solvents which
prevents new lipids biosynthesis. The lipids are extracted into the solvent and then subjected to TLC.
The lipids can be labeled in three ways. Some will be labeled with 32 P alone, some with
TNBS alone, and some with both 32P and TNBS. TLC (or other techniques such as HPLC or GC) can easily separate PC
and TNBS-labeled PC since they have different structures and hence will migrate to different places on a TLC plate.
No routine chromatography technique could, however, separate PC and 32 P-PC, since their molecular structure is
the same, the only difference being in the nuclei of the P (different number of neutrons).
Those lipids with double labels (TNB and 32 P) must have flipped from the inner leaftlet to
the outer leaftlet where they could be labeled with TNBS. The cells incubated for 3 minutes before the addition of TNBS have
a much higher level of doubly labeled PL's. Quantitating these data as a function of differing time of incubation at elevated
temperatures show that the rate of flip-flop diffusion is much higher in cells than liposomes, which suggests that the process
is catalyzed by an enzyme (flipase) in cells.
Figure: Flip/Flop diffusion in bacterial cells A: Labeling inner leaflet phospholipids
with 32 P
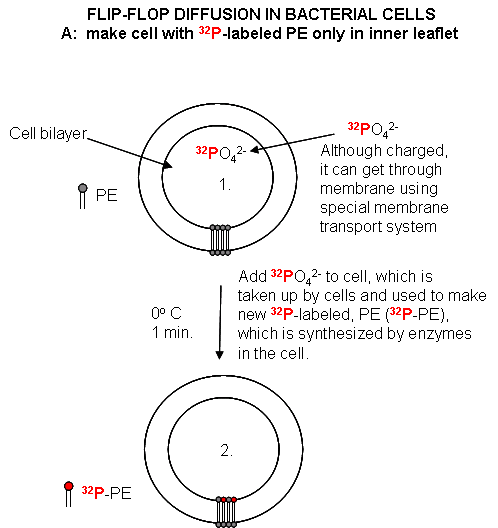
Figure: Flip/Flop diffusion in bacterial cells B: Labeling
Cells in A with TNBS to detect Flip/Flop
Lipids in membranes are often distributed asymmetrically.
The inner and outer leaflet of a biological membrane usually have different PL compositions. For example, in red blood
cell membranes, the outer leaflet is composed mostly of sphingomylein (SM) and PC, while the inner leaflet it composed mostly
of PE and phosphatidyl serine (PS). This phospholipid contains the amino acid serine linked through its side chain (-CH2OH) to phosphate in position 3 of diacylglyerol.
With a negative charge on the phosphate and carboxylate and a positive charge on the amine of PS, this phospholipid is acidic
with a net negative charge. In fact, all the PS is located in the inner leaftet! This observation will become
important latter on, when we discuss programmed cell death. A dying cell will expose PS to the outside. This is
in fact one of the markers of a dying cell.
The membrane lipid composition in an average mammalian cell
| Lipid |
% |
| PC |
45-55 |
| PE |
15-25 |
| PI |
10-15 |
| PS |
5-10 |
| PA |
1-2 |
| SM |
5-10 |
| cardiolipin (bis-PG) |
2-5 |
| cholesterol |
10-20 |
In addition, lipids of a given type often cluster within a leaftlet to form lipid "rafts"
which is analogous to a phase separation. Divalent cations like calcium, which can bind to negatively charged PLs like
PS, can cause "rafts" of PS to form, which is an example of lateral asymmetry within a leaftlet of a bilayer. Rafts
also appear to be enriched in cholesterol and lipids with saturated fatty acids, which would lead to regions of enhanced packing
and reduced fluidity. Rafts probably bind or exclude binding of other biological molecules like proteins.
Recent studies have shown that the Ebola virus interacts with lipid rafts in the process of entering and exiting the infected
cell. Rafts are also involved in how cells sense and respond to their environment. Signaling molecules on
the outside of the cell can bind receptor proteins in the membrane. As we will see later, conformational changes in
the receptor protein signals the inside of the cells that the receptor is bound with a ligand. Once bound, the receptor
can move in the membrane and often cluster in outer leaflet rafts that contain cholesterol and sphingholipids. Inner
leaflet rafts have also been observed.
Lipid Conformational Transitions
If a liposome preparation is placed in a sensitive calorimeter and the temperature slowly increased, it
is observed that the liposome preparation absorbs a significant amount of heat at a temperature characteristic of the PL which
compose the liposome. This is analogous to what would happen if solid water was heated. At the melting point of water, an
increment of heat is required, the heat of fusion, to break H-bonds and cause melting. Likewise the heat of vaporization is
observed when H-bonds are broken on the liquid-gas transition. These transition are associated with non-covalent processes,
namely, breaking H-bonds.
Figure: Heat absorption and water: phase transitions
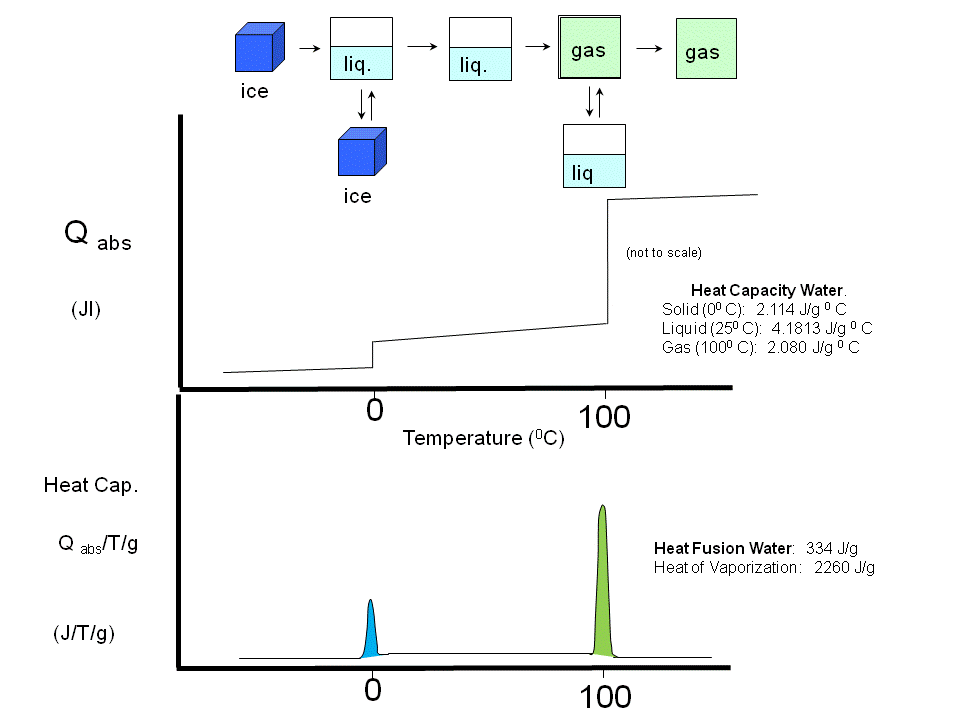
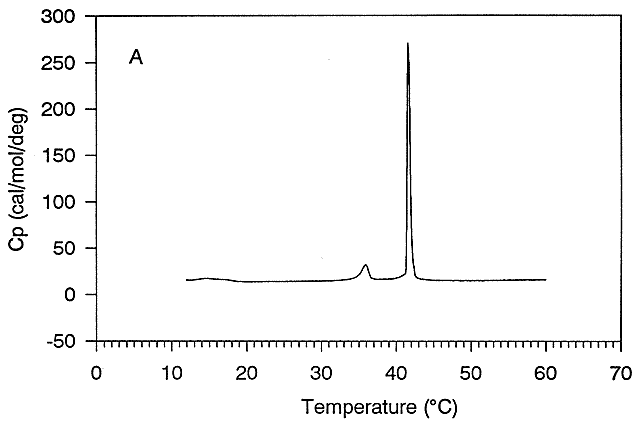
Figure: Differential Scanning Calorimeter (DSC): Phase transition for DPPC
(Dipalmitoyl phosphatidylcholine)
The transition observed with liposomes is caused by conformational chains in the packing of the acyl chains
of the PLs, as the acyl chains change from trans to gauche conformations. Before the transition, the acyl chains
are more tightly packed in the gel phase, and after they are less tightly packed in the liquid crystalline phase,
since many chains are in the gauche conformation.
Differences in Gel and Liquid Crystalline Phases in Phospholipids Bilayers
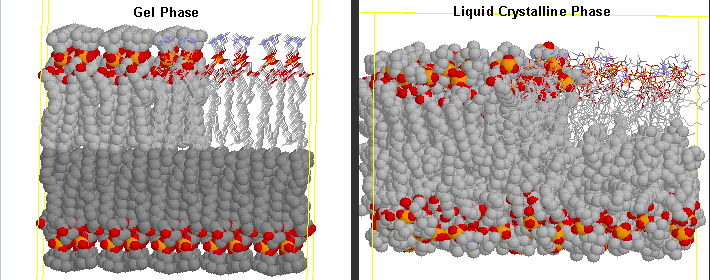
The midpoint of the phase transition is called the melting point, Tm. Vesicles in the liquid crystalline
phase are more fluid, dynamic, and hence more permeable. Note that the liposomes have not been destroyed but simply have undergone
a phase change, much like ice turning to liquid water. Liposomes made of different PL have different Tm.
Vesicles make from PLs with bigger head groups have a lower Tm, since they are less "stable". For
example, the Tm for vesicles of di-16:0 versions of PA, PE, and PC have Tm's of 67, 63, and 41 degrees C, respectively.
Cholesterol is also a ubiquitous component of animal cell membranes. Its size will allow it to fit into either leaftlet with
its polar OH pointed to the outside. One function of cholesterol in membranes is to keep the membrane fluid at any reasonable
temperature. When a membrane is at a temperature less than the Tm, it is ordinarily in a gel, not liquid crystalline phase.
The cholesterol helps prevent ordered packing of the acyl chains of the PL's, which increases their freedom of motion. Hence
the fluidity and permeability of the membrane is increased. At temperatures greater than the Tm, the rigid ring of cholesterol
reduces the freedom of the acyl chains to rotation, and hence decreases the number of chains in the gauche conformation. This
decreases the fluidity and permeability.
Large Scale Conformational Transitions
Large scale conformational transitions can occur in lipid bilayers. These include bud
formation, formation of vesicles which split off from the membrane, and fusion of membrane vesicles, all of which are important
biologically. To some extent, the tendency to engage in these larger shape transitions depends on the local curvature
of the vesicle, which depends on the local phospholipid composition and fluidity of the membrane. We have previously
seen that segregation of lipids into rafts (or domains) within a leaflet depends on the phospholipid content. Baumgart
et al. have made giant unilamellar vesicles (GUV) containing a mixture of three lipids: sphingomyelin (SM), dioleylphosphatidylcholine
(DOPC), and cholesterol (C) to study transitions in the bilayers. SM and C segregate laterally into a "liquid phase"
domain characterized by significant order (Lo) while DOPC forms a "liquid phase" with more disorder (Ld). They could
detect the different phases through fluorescence microscopy of GUV's labeled with the fluorophores. The first, N-lissamine
rhodamine dipalmitoylphosphatidylethanolamine (red fluorescence), partitions almost exclusively
into the Ld phase. The second, perylene (blue fluorescence), partitioned
with great selectivity into the Lo phase. The results of their studies are shown in the figures
below. This images provide stunning visualization of lipid domains (rafts) including those in the shapes of circles,
stripes, and rings. Their works shows that the boundary between the domains is important in determining lipid structures.
Structures are favored which reduce the perimeter at the boundaries of the phases. It appears that the Ld phase (and
associated lipids) is found preferentially in area of high membrane curvature, while the Lo phase is found is regions of lower
curvature. the two figures below are reprinted with permission of Nature and the authors: Baumgart,
Hess, and Webb, W. Imaging coexisting fluid domains in biomembrane models coupling curvature and line tension.
Nature. 425, pg 821 (2003)
Figure: Equatorial cross sections of GUVs with multiple phases
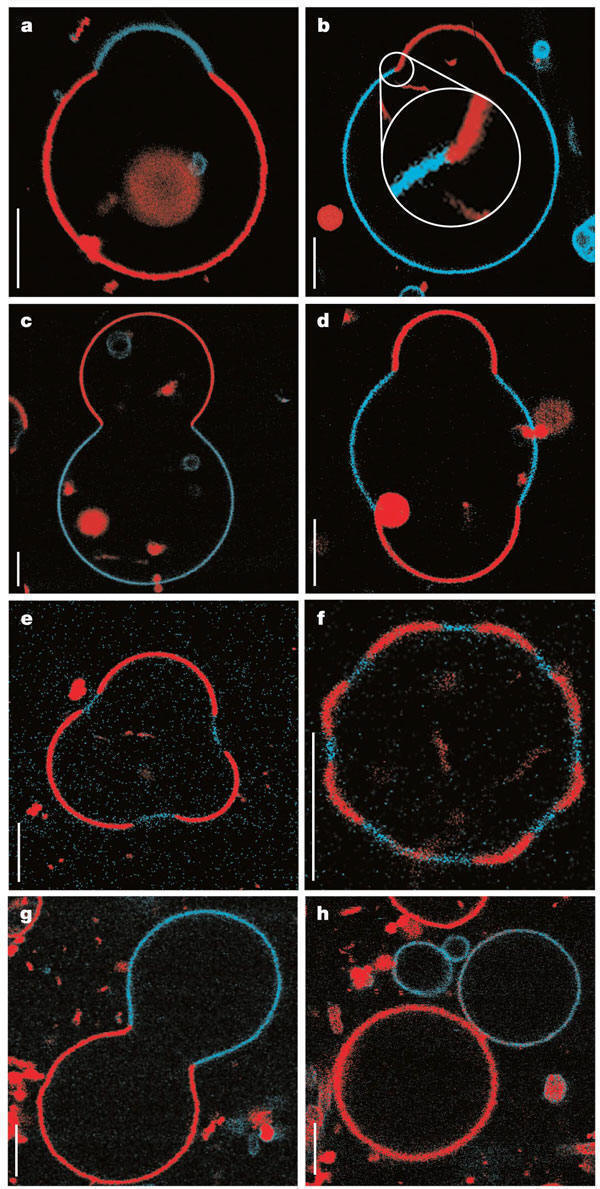
Figure: GUVs structures with multiple
phases

Membrane Permeability
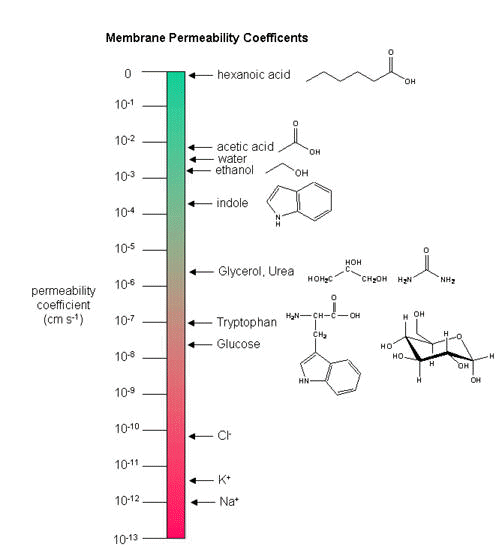
Finally, one can measure the permeability of ions and molecules across the bilayer. Permeabilities
are correlated to the partition coefficient of the molecule or ion in organic solvents. If the molecule readily dissolves
in a nonpolar solvent, it is more likely to pass through the hydrophobic barrier of the membrane. Obviously the size of the
molecule would play a part as well. Hence charged ions like Na+ and K+ have low permeabilities.
Cl- has a higher permeability, since the charge density on the larger Cl anion is smaller. Water has
a surprisingly high permeability although it is polar. It is small and can enter down deep into the head group region through
sequential H-bonding which must assist its transfer across the membrane.
Figure: Permeability of liposome bilayers
|

